
Application of EVA and Its Modified Polymers in the Field of Crude Oil Pour Point Depressants
What is a pour point depressant?
A pour point depressant (PPD) is a type of oilfield chemical additive designed to improve the low-temperature fluidity of crude oil. It works by changing the crystallization behavior of wax in crude oil, inhibiting the formation of a rigid three-dimensional network structure of wax crystals, dispersing wax crystals effectively, and preventing them from sticking together. As a result, it lowers the pour point and viscosity of crude oil, allowing crude oil to flow smoothly at lower temperatures and ensuring safe and efficient oil gathering, storage, and pipeline transportation.
Abstract
1 Traditional EVA Pour Point Depressants
1.1 Structure and Action Mechanism of EVA
As a traditional polymer-type pour point depressant, the molecular structure of EVA pour point depressant consists of two parts: non-polar long-chain alkyl groups and polar groups. Its molecular structure is shown in Figure 1. The non-polar long-chain alkyl groups, as crystallization nuclei, precipitate eutectically with wax crystals at low temperatures, inhibit the precipitation of wax crystals and reduce the total amount of precipitated wax; the polar groups are ester groups, which have a repulsive effect on wax molecules, improve the morphology and size of wax crystals, hinder the aggregation and growth of wax crystals, weaken the formation of a three-dimensional network structure of wax crystals to a certain extent, thereby dispersing wax crystals and reducing the coating of liquid oil. Finally, the synergistic effect of non-polar and polar groups achieves the purpose of reducing oil viscosity. Therefore, EVA mainly reduces the pour point and viscosity of waxy crude oil by participating in the wax precipitation process, changing the size and morphology of wax crystals, and dispersing wax crystals to inhibit the formation of a network structure.
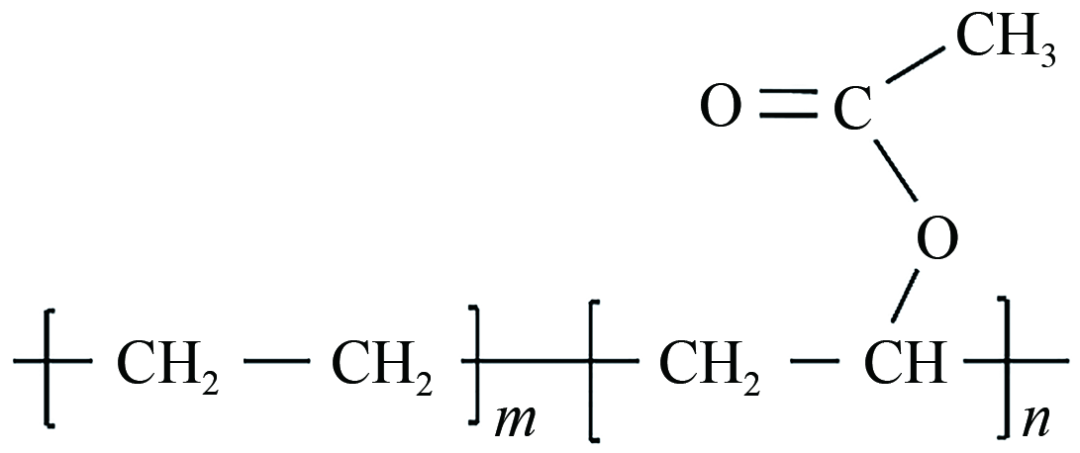
1.2 Factors Affecting the Effect of EVA
2 Chemical Modification
2.1 Graft Modification
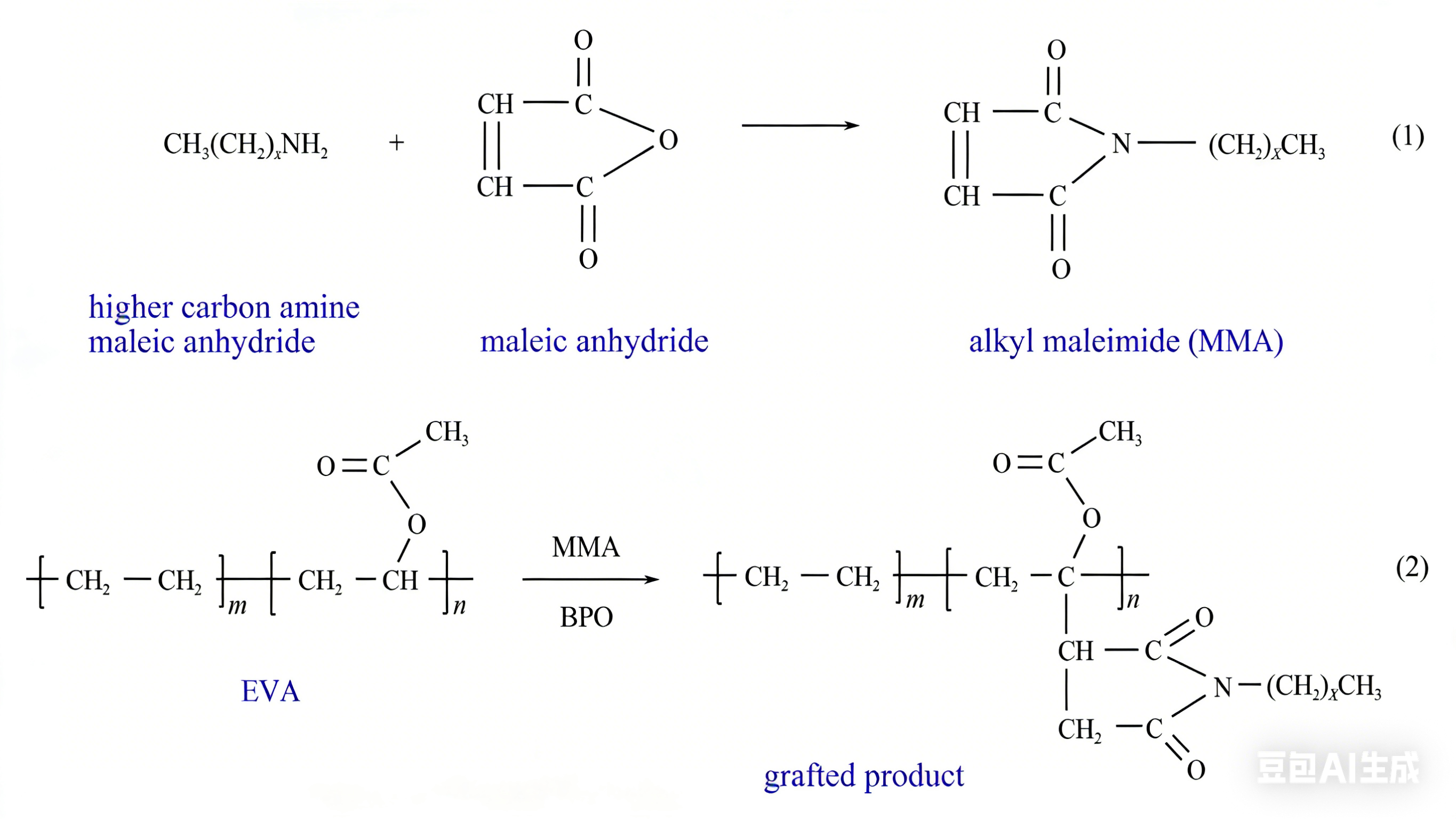
Maleic anhydride (MAH) is a commonly used chemical modification monomer. In the plastic industry production, MAH-modified EVA (EVA-g-MAH) is often used as a compatibilizer for composite materials to improve the compatibility between various components of composite materials, so as to achieve the purpose of improving the comprehensive performance of composite materials. In the field of storage and transportation, some researchers have found that introducing maleic anhydride groups into EVA molecules can enhance the polarity of EVA to a certain extent and improve its dispersion effect on wax crystals. At the same time, it can also be used as a bridge to introduce long-chain alkanes into EVA molecules to enhance its eutectic effect with wax crystals. Guan Zhongyuan et al. used dodecyl maleic secondary amide prepared by dodecylamine and maleic anhydride at a ratio of 1:1 as the graft monomer, and grafted it onto EVA (28%) by solution grafting. The research results show that this method makes EVA have longer side chains and introduces nitrogen-containing polar groups at the same time, so that the modified polymer can crystallize and precipitate wax both on the main chain and on the side chains, thereby increasing the crystallization performance of EVA. At the same time, due to the strong polarity of nitrogen-containing groups, it has the function of avoiding and resisting the connection of wax crystals to form a three-dimensional network structure, thus effectively reducing the viscosity of crude oil. It is worth noting that the carbon chain length of the graft monomer has an important influence on the graft modification effect. Wu et al. grafted alkyl maleimides, the graft monomers obtained by high-carbon amines with different carbon numbers (C₁₂, C₁₆, C₁₈) and maleic anhydride at a ratio of 1:1, onto EVA under the action of the initiator benzoyl peroxide (BPO) (the mechanism is shown in Figure 2). The grafting rate of the modified polymer decreases with the increase of carbon number, and EVA-16 has the best effect among the modified polymers, which can reduce the pour point of Daqing crude oil from 31℃ to 19.5℃.
2.2 Alcoholysis Modification
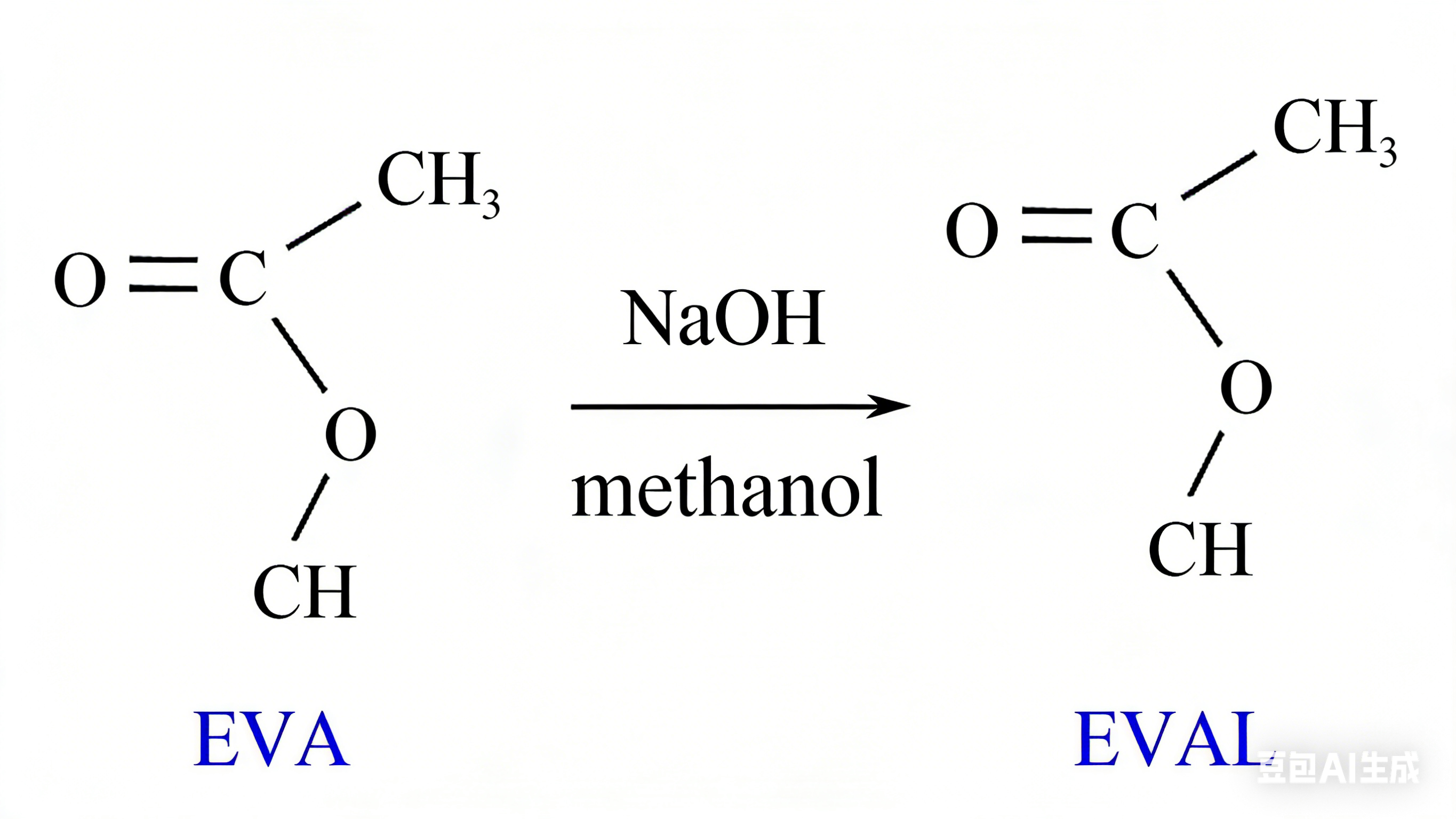
Ren et al. catalyzed with sodium hydroxide and modified EVA by methanol alcoholysis to obtain the alcoholysis product EVAL, and studied the pour point depression effect of EVAL. Compared with EVA, most of the ester groups (mole fraction 87.94%) of EVAL were replaced by hydroxyl groups (the mechanism is shown in Figure 3). EVAL with different alcoholysis degrees were prepared by changing the concentration of sodium hydroxide-methanol solution, and EVAL-1, EVAL-2, EVAL-3, EVAL-4, EVAL-5 [alcoholysis degree up to 90.94% (mole fraction)] and EVAL-6 were obtained. The research results show that both EVA and EVAL can change the wax crystallization habit and wax crystal morphology, but compared with EVA, EVAL introduces a large number of hydroxyl groups, has stronger polarity, provides greater electrostatic repulsion to wax crystals, and significantly reduces the amount of wax precipitation. The wax crystals precipitated from crude oil treated with EVAL are more difficult to connect together and form a looser network structure (Figure 4). In addition, with the increase of alcoholysis degree, the pour point depression efficiency shows a trend of first increasing and then decreasing, and the efficiency is the highest when the alcoholysis degree is 90.94% (EVAL-5), which can reduce the pour point of Jianghan crude oil by 11℃. When the alcoholysis degree is higher than 90.94%, the performance of EVAL deteriorates, but the pour point depression effect of all EVAL is better than that of EVA.
Although chemical modification can significantly improve the effect of EVA on waxy oil, polymer pour point depressants still have problems such as poor resistance to repeated heating and shear resistance at present. With the development of nanomaterial technology, the preparation of EVA/nanoparticle composite pour point depression systems by nano-hybridization provides a new path for the design and development of new and efficient EVA-based nano-composite pour point depressants.
3 Nano-hybrid Modification
3.1 EVA/Inorganic Nanoparticle Hybridization
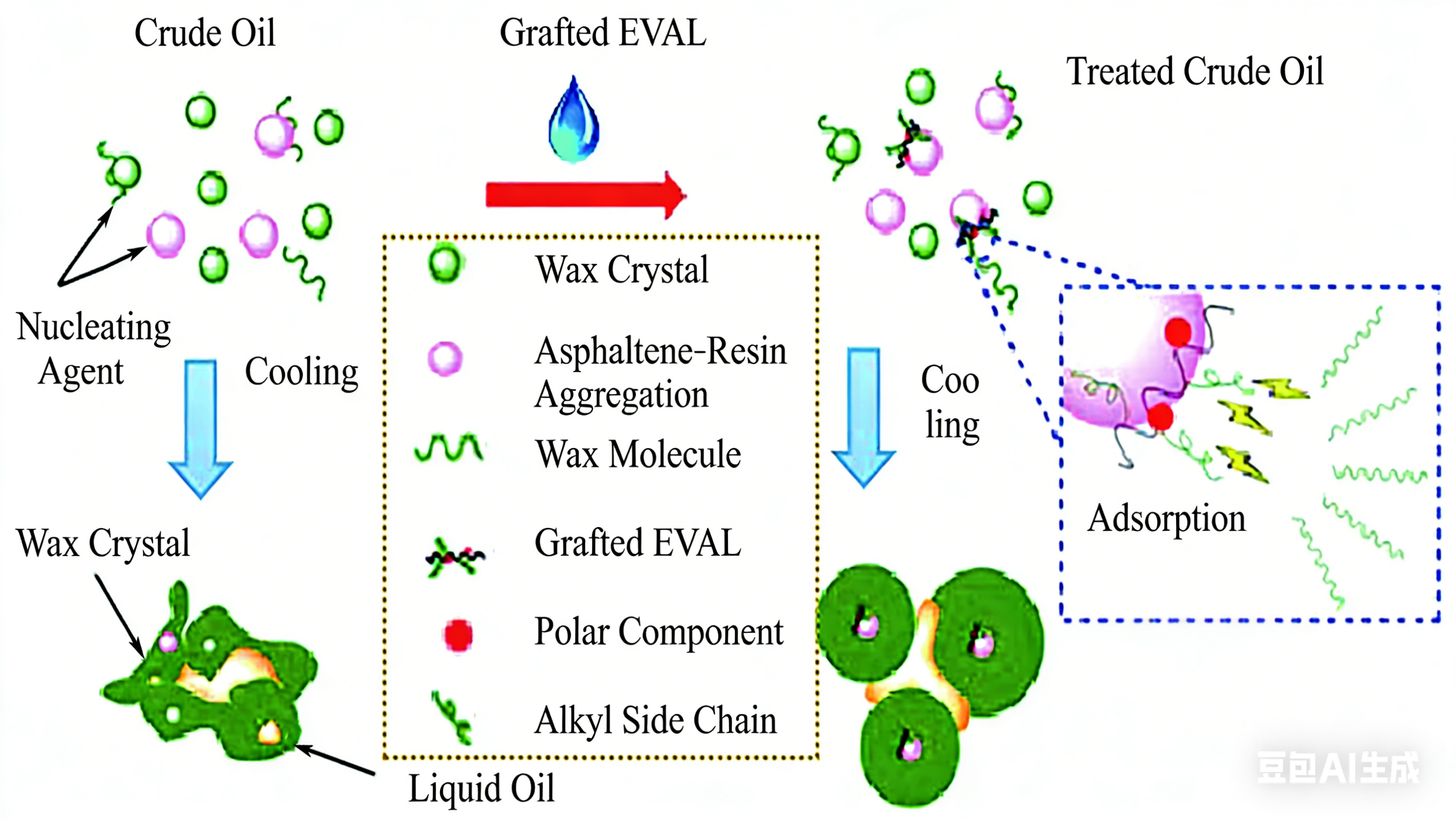
Nano-silica has high surface activity and designability because several hydroxyl groups on its surface can be replaced by other groups, and it is a commonly used nanoparticle for nano-hybrid modification of EVA. Some studies have found that the EVA/YSiO₂ pour point depressant prepared by compounding SiO₂ and EVA can make wax crystals form a compact structure through heterogeneous nucleation, and disperse wax crystals at the same time, significantly improving the low-temperature fluidity of crude oil and reducing the pour point. However, when the wax content in the simulated oil system is high, the pour point cannot truly reflect its effect on waxy oil. This is because when the wax content is high, the wax crystals of waxy oil with nano pour point depressant added diffuse and grow outward along the surface of nanoparticles. Although a certain closed space is formed between the edges of the grown wax crystals, which inhibits the flow of the oil sample, this structure is easily destroyed by shear force compared with the three-dimensional network structure formed without adding a pour point depressant, thus restoring the fluidity of waxy oil. In addition, the content of VA groups in EVA is a key factor affecting the pour point depression effect of the pour point depressant, because with the increase of VA content, the organic compatibility between the two decreases. This reduced compatibility makes EVA easier to desorb from the surface of SiO₂, and the effect of nano-hybrid PPD is mainly determined by the coverage of the polymer on the surface of nanoparticles, so too high VA content will lead to the weakening of the pour point depression effect of nano-hybrid pour point depressants. Li et al. further confirmed the influence of VA content on the effect of the prepared composite pour point depressants. They hybridized and compounded SiO₂ and montmorillonite (MMT) with EVA of different VA contents and applied them to waxy simulated oil. It was found that with the increase of VA content, the solubility of the composite pour point depressant in oil deteriorated, leading to the reduction of the effect. In addition, although the hybrid composite pour point depressant can significantly reduce the pour point of simulated oil and crude oil, its inhibitory performance on simulated oil is stronger than that on crude oil. This is because there are resins and asphaltenes in crude oil, which have large molecular weights and contain polar groups such as amino and hydroxyl groups. Due to intermolecular forces, resin and asphaltene molecules are entangled with each other, and are more likely to form gels with the decrease of temperature (the action mechanism is shown in Figure 7), which affects the effect of the composite system.
In addition to morphology and structure, some nanoparticles also have their own functional properties, such as magnetism. Yu et al. hybridized magnetic Fe₃O₄ and non-magnetic SiO₂ with the same morphology, structure and size with EVA respectively and applied them to Daqing crude oil. The pour point depressant hybridized by Fe₃O₄ and EVA is denoted as F-NPPD. When a small amount of F-NPPD is dispersed in the oil phase, with the decrease of temperature, F-NPPD first co-crystallizes with wax crystals according to the heterogeneous nucleation mechanism, and the own magnetism of F-NPPD will further induce the stacking of wax crystals on its surface. With the gradual weakening of the outer magnetism, the wax crystals finally form a petal-like structure (the mechanism is shown in Figure 8). The experimental results show that compared with the S-NPPD pour point depressant hybridized by SiO₂ and EVA, the F-NPPD pour point depressant can better induce wax crystals to form an ordered compact structure through its own magnetism, inhibit the formation of gel structure and reduce the viscosity of crude oil. Some studies have further confirmed that compared with pure EVA or EVA hybridized with non-magnetic nanoparticles, the hybridization of magnetic nanoparticles with EVA can effectively change the precipitation mode of wax crystals, increase the dispersion degree of wax crystals and make the interior more compact, thus reducing the yield stress of crude oil. After applying an alternating magnetic field with an intensity of 0.3T and a frequency of 20Hz to the system, the yield stress is further reduced and the wax crystals are looser. However, increasing the intensity and frequency of the alternating magnetic field will cause the wax crystals to aggregate to a certain extent, resulting in a rebound of the yield stress. Peng Zeheng et al. studied a linear nano pour point depressant. Applying an alternating magnetic field of 0.3T and 20Hz to it can significantly reduce the yield stress of crude oil, but applying a constant magnetic field will cause a rebound of the yield stress of crude oil. This is because under the action of a constant magnetic field, the three-dimensional network structure formed by the eutectic or heterogeneous nucleation of wax molecules and pour point depressant molecules re-aggregates to a certain extent, which strengthens the strength of the three-dimensional space network structure, thus leading to the enhancement of yield stress. In addition, compared with EVA, the linear nano pour point depressant has a more obvious effect on changing the morphology and aggregation degree of wax crystals due to its unique structure, because it is a linear nano-long chain with a large specific surface area, which can graft more polymer molecules on the surface, enhance its interaction ability with wax crystals and further change the network structure of wax crystals, thus making the effect of reducing yield stress stronger.
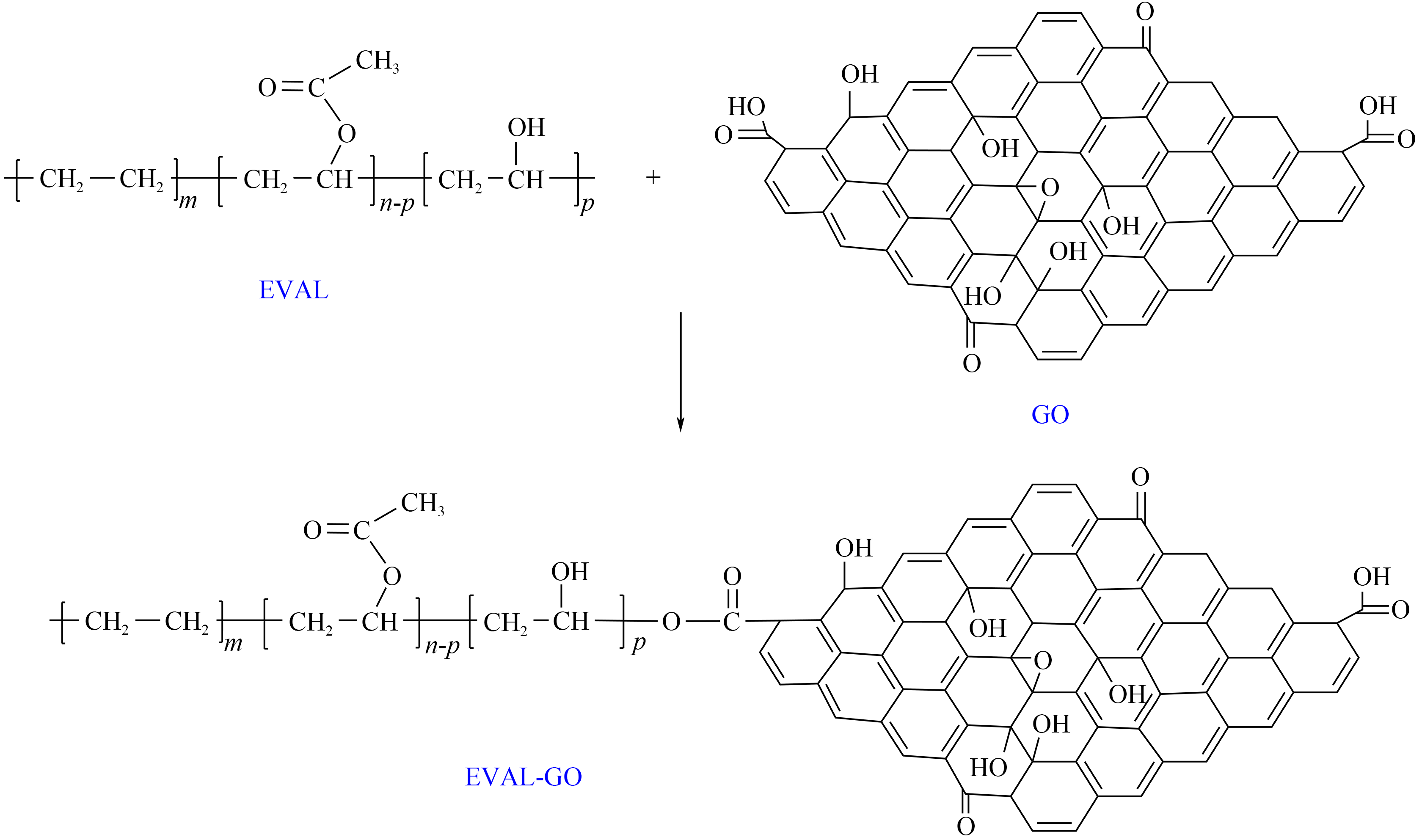
The compatibility between EVA and inorganic nanoparticles is the key factor restricting the effect of their hybrid composite system. In recent years, some researchers have found that nano pour point depressants prepared by hybridizing carbon-based nanoparticles (such as graphene, carbon nanotubes) with polymers have better stability in the oil phase. Using the functional groups on the surface of carbon-based nanoparticles, the stability and pour point and viscosity reduction effects of the prepared nano-composite pour point depressants can be further improved through in-situ polymerization or chemical grafting. Liu et al. first prepared EVAL by alkali-catalyzed alcoholysis, and then prepared EVAL-GO nano-composite pour point depressant through esterification graft reaction between the carboxyl groups on the surface of graphene oxide (GO) and the hydroxyl groups on EVAL (the mechanism is shown in Figure 9). Compared with the nano-composite pour point depressant prepared by simple physical adsorption, the nano-composite pour point depressant prepared by grafting has significantly improved stability in the oil phase, so that it can not only inhibit the precipitation of wax crystals, but also improve the dispersibility of wax crystals, turn wax crystals into spheres and increase the flow space of liquid oil. At the same time, it can significantly reduce the pour point, gel point, yield stress and viscosity of crude oil, thus changing the low-temperature fluidity of oil.
3.2 EVA/Organic-inorganic Nanoparticle Hybridization
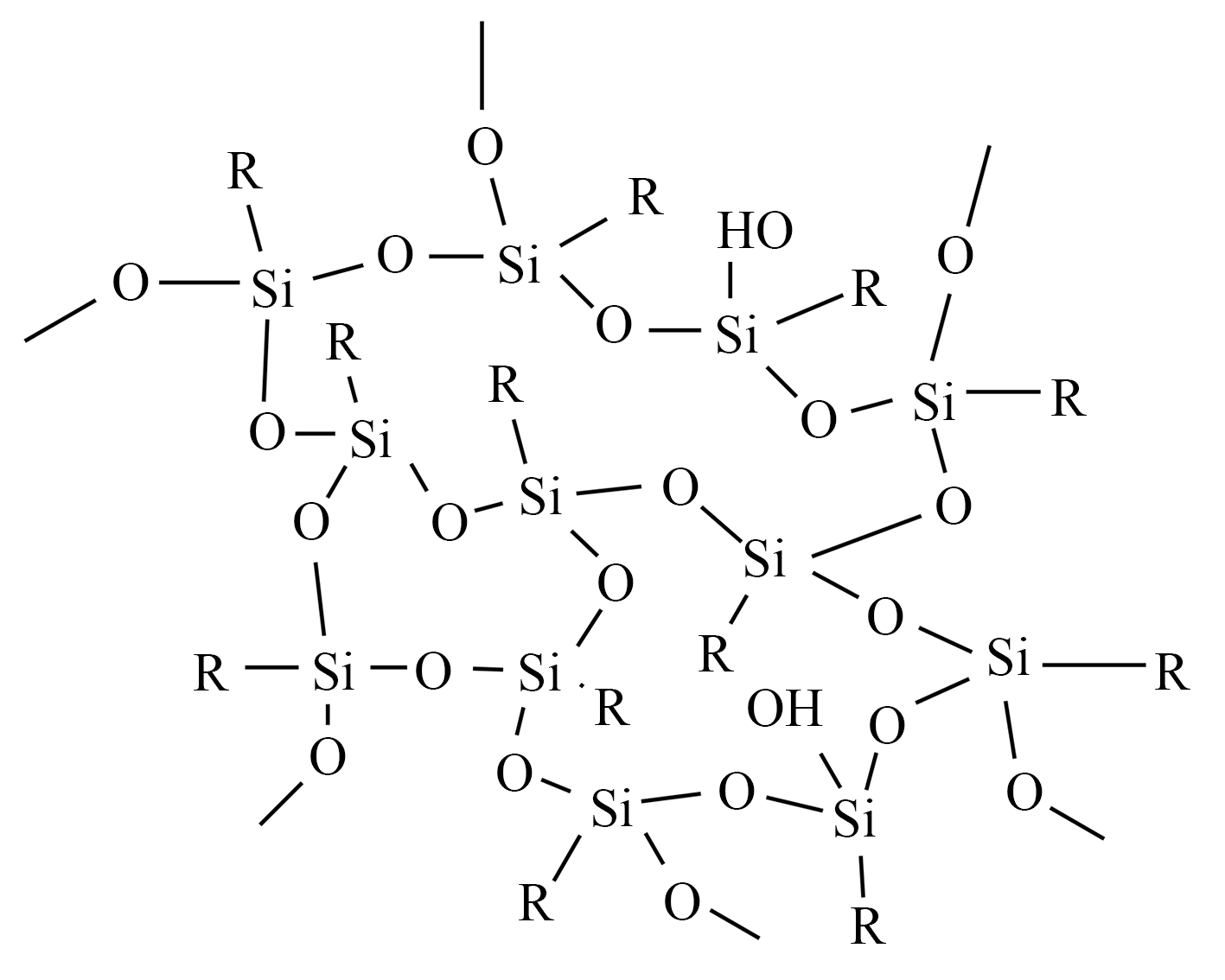
Polyorganosilsesquioxane (PSQ) microspheres, an organosilicon material (the structure is shown in Figure 10), have excellent morphological and structural properties. PSQ refers to an organosilicon polymer with the structural formula (RSiO₃/₂)ₙ, where R can be hydrogen, alkyl, aromatic, olefin groups and organic group substituents. The molecular structure of PSQ contains both an inorganic siloxane "skeleton" structure and a variety of organic functional groups can be introduced through side groups, which has both organic and inorganic properties, and has good thermal stability and mechanical properties without organic modification. At present, the sol-gel two-step method is commonly used to prepare PSQ microspheres with low cost. PSQ microspheres usually have a regular spherical morphology and can be monodispersed in the oil phase, so the use of PSQ to prepare nanocomposites has become a research hotspot.
Yao et al. prepared polymethylsilsesquioxane (PMSQ) microspheres by the sol-gel two-step method, introduced methyl groups into PSQ, and compounded the two by melt blending to form EVA/PMSQ composite particles. The pour point depressant can be used as a heterogeneous nucleation template for wax precipitation, increasing the size and density of wax crystals, thus further improving the low-temperature fluidity of crude oil. The adsorption amount of the pour point depressant on the surface of microspheres is the key factor determining the pour point depression effect. However, a large number of methyl groups are introduced into PMSQ, although its dispersibility in oil is improved, it will affect the adsorption amount of EVA in microspheres. With the increase of microsphere concentration, the adsorption amount of EVA will decrease significantly, thus leading to the deterioration of pour point depression performance. On this basis, Yao et al. prepared polyaminopropyl/methylsilsesquioxane (PAMSQ) microspheres by the sol-gel two-step method, and compounded PAMSQ with EVA by melt blending to form EVA/PAMSQ composite particles (the action mechanism is shown in Figure 11). Compared with PMSQ, PAMSQ introduces amino groups on the basis of PMSQ. Although its oil solubility is deteriorated, its adsorption amount of EVA is much larger than that of PMSQ for EVA, and the high surface energy caused by the morphological defects on the surface of PAMSQ further increases the adsorption amount of EVA, thus making the pour point depression effect of the composite pour point depressant more significant. Compared with EVA/PMSQ, EVA/PAMSQ provides a stronger heterogeneous nucleation template for wax crystals, making the wax crystals larger in size and stronger in aggregation degree, thus further improving the low-temperature fluidity of crude oil, significantly reducing the apparent viscosity, yield value and gel point of crude oil, and improving the low-temperature fluidity of crude oil. However, the introduction of amino groups needs to be appropriate. Excessively high amino concentration will lead to the oil solubility of microspheres, and ultimately reduce the pour point depression effect of the composite pour point depressant.
4 Synergistic Modification
4.1 Physical Synergism
Physical synergism is the process of enhancing the effect of EVA on waxy oil by applying an external electric or magnetic field. Researchers have found that placing waxy oil under a high-voltage electric field can improve its cold fluidity. Xie et al. combined EVA with an electric field to treat waxy crude oil, and verified whether the pour point depressant under the action of a combined electric field has a better effect than treating waxy crude oil with EVA alone or electricity alone. The research results show that compared with single electric treatment and EVA treatment, the combined treatment can more effectively reduce viscosity, weaken waxy gel and reduce thixotropy, and can broaden the size distribution of wax crystals and shift it to a larger average particle size distribution, thus improving the cold fluidity of waxy crude oil. Huang et al. pointed out that not only electric treatment can reduce the viscosity of waxy crude oil, but also magnetic treatment can effectively reduce the viscosity of crude oil. Therefore, they studied the effects of the intensity and frequency of alternating magnetic field on the yield stress of waxy simulated oil with/without EVA pour point depressant and EVA/nano-MMT pour point depressant (NPPD) (shown in Figure 12). The research found that: ① After magnetic field treatment, wax crystals continue to aggregate, the liquid oil coated in the three-dimensional network structure of wax crystals is released, and the morphology of wax crystals also changes, resulting in the reduction of yield stress; ② The synergistic effect of EVA and magnetic field is better than that of EVA alone or magnetic field treatment alone, and the same conclusion is obtained for the synergism of NPPD and magnetic field, and the pour point depression effect is stronger than that of the synergism of EVA and magnetic field; ③ There are optimal values of intensity and frequency for the treatment of waxy crude oil by magnetic field synergistic pour point depressants. For the EVA and magnetic field synergistic system, the optimal values are 0.4T and 40Hz; for the NPPD and magnetic field synergistic system, the optimal values are 0.3T and 20Hz. For any system, when the magnetic intensity or frequency is lower than the optimal value, the yield stress decreases with the increase of intensity and frequency. However, when the magnetic field is further increased after the optimal value, the aggregation of wax crystals will change, and the construction degree of the three-dimensional network structure of wax crystals will be enhanced, thus weakening the pour point depression effect.
4.2 Chemical Synergism
Chemical synergism is the process of adding EVA and other chemical components to the waxy oil system at the same time to synergistically improve the low-temperature fluidity of waxy oil. Crude oil is a complex mixture containing not only wax and resins, but also asphaltenes. From the perspective of elemental composition and physical and chemical properties, the non-hydrocarbon component with the largest molecular weight, the strongest polarity, the most heteroatoms and the highest metal content in petroleum is defined as asphaltene. Asphaltene is the most polar part in crude oil, can be dispersed in the oil phase as aggregates, and is considered a natural pour point depressant. As a natural pour point depressant, asphaltene can synergistically improve the rheological behavior of waxy crude oil with polymer pour point depressants. Yao et al. studied the synergistic improvement of the flow behavior of simulated waxy oil by EVA and asphaltene, and studied the effects of wax content, asphaltene content and VA content of EVA on the synergistic effect of EVA/asphaltene composite pour point depressant respectively, and obtained the following conclusions: ① Both EVA and asphaltene can improve the low-temperature fluidity of crude oil to a certain extent, but the synergistic use of EVA and asphaltene has a better effect; ② Asphaltene is usually dispersed in crude oil in the form of associated colloidal particles (micro-nano scale), while resins are adsorbed on the surface of asphaltenes or interspersed in the asphaltene structure as stabilizers to stabilize the dispersion of asphaltenes. EVA molecules can be adsorbed on the surface of asphaltene aggregates stabilized by resins, thus inhibiting asphaltene precipitation and forming EVA/asphaltene composite particles. As a wax crystallization template, it changes the morphology of wax crystals into large, compact and spherical wax crystal flocs, thus significantly improving the flow behavior of waxy oil (Figure 13); ③ With the increase of wax content, the synergistic effect deteriorates, but it still has good pour point depression performance when the wax mass fraction is 20%; ④ The synergistic effect of EVA/asphaltene composite particles on the fluidity of waxy oil first improves with the increase of asphaltene mass fraction, but there is an optimal range of 0.75%~1.5%. When the asphaltene mass fraction is low (3%), the EVA/asphaltene composite particles disperse the precipitated wax flocs into relatively small spherical waxes, increasing the solid-liquid interface area, and also overlap each other to form a three-dimensional network structure under the action of asphaltenes. Too low or too high asphaltene mass fraction will hinder the flow performance of crude oil; ⑤ The increase of EVA polarity can enhance the synergistic effect of EVA/asphaltene. With the increase of VA content, the wax crystals become more regular and dense, the shape becomes spherical, the wax crystals are not easy to overlap, and more liquid oil is released, thus improving the effect of inhibiting crude oil gelation. However, the increase of EVA polarity needs to be moderate. If the polarity is too high, it is not conducive to eutectic crystallization and will deteriorate the pour point depression effect. In addition, surfactants are also often used as crude oil pour point depressants, and their single use effect is poor, so they are often used synergistically at home and abroad. Fluorine-containing surfactants can improve the pour point depression effect. Yu Hongjiang et al. studied the synergistic effect of fluorine-containing surfactants and EVA on waxy crude oil and found that the above pour point depressants can improve the morphology of wax crystals, turn acicular and rod-like wax crystals into spheres, reduce the solid-liquid interface energy in crude oil, thus greatly reducing the pour point of crude oil. Studies have found that gossypol is a polyphenolic compound containing many polar and non-polar groups, which is easily soluble in oil. Therefore, Marenov et al. put forward a bold hypothesis to use the composition of EVA and crude gossypol as a composite pour point depressant. The results show that the composite pour point depressant has a better pour point depression effect than pure EVA, can further prevent and destroy the growth of wax crystals, and prevent wax crystals from connecting to form a three-dimensional network structure.
| Modification method | Purpose | Influencing factors | Advantages and disadvantages |
|---|---|---|---|
| Chemical modification | Introduce polar groups into EVA molecules to enhance their dispersion effect on wax crystals, or introduce long-chain alkanes to enhance their eutectic effect with wax crystals | Selection of graft monomers, alcoholysis degree | Improve the pour point depression performance of EVA from the perspective of molecular structure design, but there are still problems such as poor resistance to repeated heating and shear resistance |
| Nano-hybrid modification | Introduce nanoparticles into the EVA matrix to change the wax crystal structure and disperse wax crystals through heterogeneous nucleation | Type, structure and properties of nanoparticles; preparation method | Significantly improve the pour point and viscosity reduction effects of EVA, with good thermal stability and shear resistance, but the influence of nanoparticles on the subsequent processing of crude oil is not clear |
| Synergistic modification | Further improve the eutectic and dispersion effects of EVA on wax crystals through synergistic effect | Synergistic medium | Physical synergism is restricted by equipment and energy consumption; the thermal stability and shear resistance of chemical synergism are not clear |